How to Measure Color
Proper analysis is key for creating plastic parts that require accurate & consistent coloration
Previous Article Next Article
By Ron Beck
R&D Manager, Americhem, Cuyahoga Falls, Ohio, USA
How to Measure Color
Proper analysis is key for creating plastic parts that require accurate & consistent coloration
Previous Article Next Article
By Ron Beck
R&D Manager, Americhem, Cuyahoga Falls, Ohio, USA
How to Measure Color
Proper analysis is key for creating plastic parts that require accurate & consistent coloration
Previous Article Next Article
By Ron Beck
R&D Manager, Americhem, Cuyahoga Falls, Ohio, USA

Spectrophotometry is one method for repeatable, accurate color matching.

Spectrophotometry is one method for repeatable, accurate color matching.
"If you choose to compare colors visually, know that the methodology you select can affect color measurement outcomes."
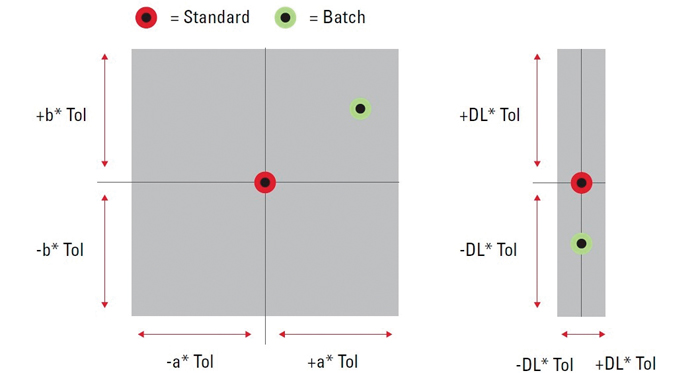
Figure 1: A “pass/fail” box, centered upon the color values of a standard, shows the area within which a sample batch value must fall.
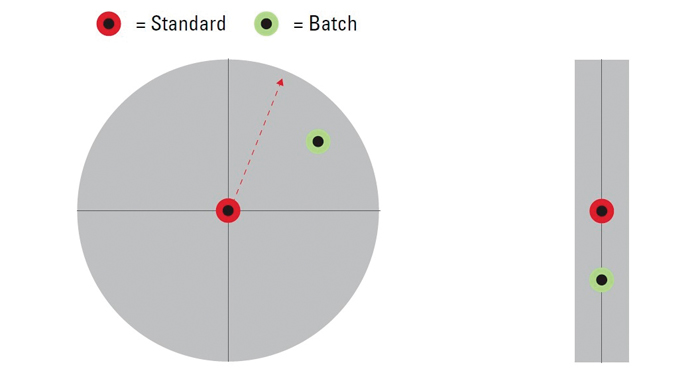
Figure 2: A circle centered upon the color values of a standard graphically shows the “spectral distance” between the standard and a sample batch.
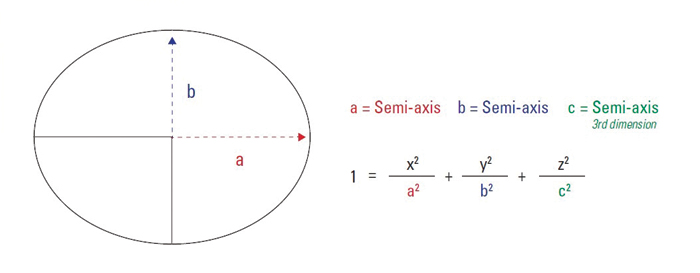
Figure 3: An elliptical pass/fail equation-based system places a customizable emphasis on hue and chromaticity
As children, one of the very first concepts you likely learned was that of “color.” As a toddler, you learned to tell red from blue. As you grew older, you began to comprehend that there is far more diversity in colors than you had originally grasped. Sea green and forest green may both be green, you found; yet there is an easily perceptible distinction between the two.
For many people, education on the differentiation of colors progresses little after childhood. They may recognize that there is a difference between two shades of a color, but they never learn how to measure this difference. And they are widely alright with this fact, as the ability to visually perceive a difference in color is what matters most to a layperson. For those who make regular decisions regarding the coloration of products, however, the skills to empirically differentiate colors are far more valuable.
So how should we measure color?
Principles of Examination
As is the case with many scientific endeavors, measuring color involves examination followed by analysis. Thorough examination requires clear procedures, responsible methods, and a firm understanding of variability. You need good representative samples with which to work, as well as statistically valid, clean, appropriately preserved standards. Any sample being measured must be statistically representative of the entire batch, free from contamination, the same form as a target, and reproducible within acceptable limits of repeatability.
Visual examination
Evaluation of color can be—and generally is—done visually, but there is a fundamental issue with this method. Perceptions of color are highly subjective, as visual examination involves many complicating variables—from differences in the eyes and brains of examiners to the conditions of the examining environment. Light sources vary in brightness and color rendering ability, for instance, and the properties of an object being examined could shift over time. As a result, consistent judgment is difficult to come by, and repeatability and reproducibility of color measurements determined visually are generally low.
If you choose to compare colors visually, know that the methodology you select can affect color measurement outcomes. For instance, if you measure a single sample (imagine a square painted with a sample of a batch of yellow paint) versus a single standard (imagine a square painted with a sample of a batch of yellow paint previously determined to be of acceptable coloration), you may find that your judgment of similarity between the sample and the standard shifts over time, making repeatability difficult. As an alternative, if you choose to compare a single standard against numerous reference batches of a color, these previous batches can provide visual anchors to help you reproduce the same judgments from examination to examination.
Instrumental examination
The alternative to visual examination is to use specialized instruments to measure color. Instrumental examination allows you to assign quantifiable values to colors, making comparison simpler and eliminating some of the inherent inconsistencies of visual judgment. Remember, however, that a machine is not a human eye, and thus the consistency of instrumental measurement should always be paired with an understanding that regardless of its numerical values, an object’s color could still appear differently depending on who is looking at it and where.
While color-identifying instruments are very useful for scientific color measurement, there are a few things to keep in mind when using an instrumental approach. First, know your instrument. An advantage that eyes have over instruments is that you know exactly how to use them. Complex instruments, however, could easily have a misadjusted setting that confuses your measurements if you are not intimately familiar with the mechanism’s workings. Ensure that whatever equipment and procedures you use will deliver results within acceptable error tolerances, and that you know how precisely your instrument will measure. Account for angle of viewing and the reflective properties of any gloss or coatings that could confound your measurements as well.
Principles of Color Analysis
Once you’ve determined the methodology you will use for color examination and have your measurements, it’s time to analyze your findings. The biggest key for color analysis is to remember that perceptibility is not the same as acceptability. Perceptibility of the difference between color samples is determined by whether they look different to you; acceptability of the variance between the two samples is established by values set in place by tolerances that vary from application to application.
In some cases, perceptible variations may be deemed acceptable based on the governing parameters of an agreement, and discarding a batch of materials based on visual analysis alone could be a costly mistake. On other occasions, depending on visual perception of color variance could mean shipping a batch of product that does not meet specifications. To avoid these issues, focus on computing whether the variance between two colors falls within a set tolerance rather than whether you can see a difference.
Establishing color tolerances
When analyzing the acceptability of a color batch, you will be scrutinizing values that portray nuanced variations between color samples. Instrument readings will convey information such as:
- ∆L: the difference in lightness/darkness
- ∆A: the difference in red/green values
- ∆B: the difference in yellow/blue values
- ∆C: the difference in chromaticity
- ∆E: the spectral “distance” between the two colors
- ∆H: the difference in hues
These values are used to determine graphically whether a batch “passes” or “fails” based on specified tolerances.
There are a number of ways to determine whether a batch falls within an acceptable tolerance, but not all methods of designating a tolerance are equally advisable. One option, for instance, is to create a “pass/fail” box centered upon the color values of the standard against which you are measuring (Figure 1). The box shows the area within which a batch must fall on the graph. If the value is within the box, it is likely to be acceptable, but not in all instances. The box assumes a linear, uniform behavior of color space, which is not an accurate assumption.
Another method is to use a circle centered upon the color values of the standard to determine whether the spectral distance between the standard and batch is small enough to fit within the circle, and therefore be deemed acceptable (Figure 2). However, as in the option of the square box, it assumes linear, uniform behavior of color space, which is not accurate.
The third option is to create an elliptical pass/fail system based on an equation set forth by the Color Manufacturing Council (Figure 3). This system places a customizable emphasis on hue and chromaticity value, allowing you to determine how much weight is placed on the hue and chromaticity differences of the two colors. The ability to adjust hue and chromaticity ratios in your calculations is important because the area of the color space in which your colors fall can impact how perceptible hue and chromaticity variances will be, and thus it’s beneficial to weigh variance in neutral and high chromaticity colors differently. Commonly used ratio values are 2:1 to determine acceptability.
A recommended approach
Creating an exact match between two colors with no visual difference is both difficult and expensive. Mathematical difference and perceptible difference are two different things, and what really matters is not whether there is variance between two colors, but whether that difference is visually close enough to be acceptable by a customer or consumer.
Of the three options, the elliptical pass/fail system gives you the best point of reference when determining a tolerance that accurately reflects whether a batch will meet this criterion. When charting and comparing real-world results of batches that were either accepted or rejected, we find that pass/fail boxes and circles designate as failures a large number of batches that were actually deemed acceptable. If you choose the elliptical pass/fail system, your probability of false negatives and false positives drops dramatically compared to the box or circle systems. The ellipse, accounting for the realities of how we view color in a non-linear, non-uniform space, offers the best chance of saving you time and money.
Some instruments calculate according to the CMC l:c (Color Measurement Committee lightness:chroma) equation used to create pass/fail ellipses; in other cases you can run the calculations yourself based on the delta values your instrument reports. For the most part, you can rely on your equipment to do the math. The responsibility to choose the right methodology and tolerance system lies with you, however, so remember your basics.
Color measurement, to some extent, is a mixture of science and art. This article offers a high-level overview of the subject to help you establish an elementary understanding of color measurement, but there is far more to be said on the topic. Even long-time color measurement veterans often have questions on the appropriate ratios and equipment to use.
About the author:
Ron Beck joined Americhem in 1976 as a product designer. After over 35 years with the company, Beck has held multiple positions. Currently, Beck specializes in color science, technical statistics, product design, and Six Sigma. Beck’s future goals for Americhem are to implement the company’s vDesign® program and exploit its new capabilities to change the business model for color concentrates, as well as to utilize his Six Sigma Black Belt training to improve plant efficiency. He can be reached at rbeck@americhem.com or +1 330-354-3346.

