Warding Off Infection
Plastics have been recruited as front-line troops in the battle against dangerous hospital-acquired infections
By Jon Evans
Previous Article Next Article

Evonik’s Cyrolite acrylic-based polymer is said to fight off infectious bacteria in parts like connectors, spikes, check valves, and filter housings.


Eastman’s Tritan copolyester is reportedly a good fit for Pevco’s clear pneumatic tube carriers used for transporting materials such as drugs and laboratory specimens around hospitals.

Being admitted to a hospital is stressful and traumatic enough as it is, without the fear that you might contract a life-threatening infection once there. But HAIs (hospital-acquired infections or healthcare-associated infections) are a very real danger.
A report published in July 2013 by the European Centre for Disease Prevention and Control (ECDC), based on a survey of over 1,000 hospitals in 30 European countries, revealed that an estimated 3.2 million people contract HAIs in Europe every year. “The survey confirms that healthcare-associated infections pose a major public health problem and a threat to European patients,” says Marc Sprenger, ECDC director. “ECDC estimates that on any given day, about 80,000 patients, [that is] one in 18 patients in European hospitals, have at least one healthcare-associated infection.” In the USA, the Center for Disease Control (CDC) reports that there are around 1.7 million HAIs in hospitals each year.
What makes HAIs particularly dangerous is that they are often caused by antibiotic-resistant strains of bacteria, which can be resistant to as many as ten different drugs, making such infections very difficult to treat. According to the World Health Organization, antibiotic-resistant strains of bacteria such as Escherichia coli, Staphylococcus aureus, and Clostridium difficile are responsible for up to 60% of HAIs in the USA.
Understandably, such antibiotic-resistant strains are most prevalent where there is widespread use of antibiotics, such as in hospitals, which also house the sick, frail, and elderly who are least able to fight off such infections. The ECDC estimates that HAIs cause 25,000 deaths a year in Europe, while the CDC has reported that HAIs contribute to 100,000 deaths a year in the USA, making them the fourth leading cause of death. If you’re sick, then the hospital is often the last place you want to be.
Efforts are now underway to curtail the spread of HAIs, with financial penalties beginning to be imposed on hospitals with high rates of infection in countries such as the USA. Hospitals are implementing strict hygiene and cleaning regimes, requiring doctors and nurses to regularly wash their hands with antimicrobial soap, and for medical instruments and devices to be sterilized before use. This is especially important for devices such as implants and catheters that are inserted into the body, because this obviously greatly increases the risk of infection.
Recently, however, another front has opened up in the battle against HAIs, as producers of plastic resin begin to enter the fray. Many medical devices are made of plastic, and the producers of these medical plastics are beginning to introduce resins that actively assist in reducing the incidence of HAIs. Indeed the development of such resins is one of the hottest topics in medical plastics at the moment, says Len Czuba, president of Czuba Enterprises, a U.S. medical polymer design company, and a leading member of the SPE’s Medical Plastics Division.
Microbes’ Enemies
The most direct way in which plastics producers are helping to reduce the incidence of HAIs is by developing plastic resins with antimicrobial activity, allowing medical devices and instruments made from these resins to restrict the growth of and/or kill pathogenic bacteria. Several companies have introduced such resins over the past couple of years, all of which take advantage of the natural antimicrobial abilities of silver nanoparticles, known as nanosilver.
In 2012, BASF introduced its HyGentic range of antimicrobial resins. This range includes: HyGentic SBC, a transparent injection-moldable styrene butadiene block copolymer that can be used to manufacture medical devices such as inhalers and ventilation filters; HyGentic PA, a glass fiber-reinforced, injection-moldable polyamide that is reportedly well-suited to producing the operating elements in medical devices; and HyGentic NW, a synthetic, non-woven fiber said to be ideal for producing surgical masks, drapes, and gowns.
BASF claims that its HyGentic range possesses excellent antimicrobial effects, preventing pathogenic bacteria such as the now infamous methicillin-resistant S. aureus (MRSA) from colonizing plastic surfaces. “BASF has developed a broad range of solutions for medical device and equipment surfaces that durably prevent hazardous microbial build-up,” says Stephen Zlock, business development manager at BASF Medical Innovation. “Polymers and elastomers featuring our additives support the measures of professional hospital hygiene. This minimizes the risk that medical devices manufactured from these products will contribute to microbe propagation.”
Also in 2012, Evonik launched Cyrex Protect, an acrylic-polycarbonate alloy, and Cyrolite Protect, an acrylic-based multipolymer. Both these plastics reportedly have proven antimicrobial activity against a variety of bacteria found in hospitals, including S. aureus, Pseudomonas aeruginosa, Staphylococcus epidermidis, and Klebsiella pneumoniae. According to the company, these plastics are particularly well-suited for producing medical equipment such as connectors, spikes, check valves, and filter housings.
“In recent years there has been interest in adding antimicrobial additives to medical devices to help reduce or prevent device-related infection,” says Peter Allread, general manager, acrylic polymers, at Evonik. “Cyrolite and Cyrex Protect have been specifically developed to address this challenge facing hospitals and clinics today.” Next year, Evonik plans to unveil the latest additions to the two product ranges, which as well as being antimicrobial will also possess outstanding chemical resistance to lipids and alcohol.
Evonik is also said to be working with the U.S. company DiFusion Technologies on developing a new antimicrobial polymer for orthopedic and dental implants. Called CleanFuze, this polymer is a combination of Evonik’s Vestakeep PEEK (polyetheretherketone) and DiFusion’s silver zeolite technology. It has been designed to contain molecular matrices of silver and zinc ions that provide both antimicrobial and bone-growth properties, meaning the polymer will not only prevent infection but will also promote bone and tissue integration. In a series of in vitro and in vivo tests in 2012, DiFusion reportedly showed that CleanFuze can eradicate 99.998% of MRSA bacteria and prevent the formation of biofilms, tough agglomerations of bacterial cells that adhere to surfaces and tend to be resistant to antibiotics.
Making similar advancements is SABIC Innovative Plastics, which launched its range of antimicrobial medical plastics in February 2013. “Intense and rising focus among regulators, hospitals, and consumers to protect patients from harm, particularly from HAIs, is putting healthcare device makers under tremendous pressure to enhance their products with antimicrobial properties and other safety attributes,” explains Thomas O’Brien of SABIC.
This new range consists of nine antimicrobial grades, which SABIC claims comprises one of the broadest antimicrobial portfolios in the industry. The nine grades cover four different product families: Lexan EXL copolymer, Lexan polycarbonate (PC) resin, Xenoy PC/polybutylene terephthalate resin, and polypropylene resins with and without fiberglass reinforcement.
Although all nine grades possess antimicrobial activity against a wide range of bacteria, fungi, and mold, they differ in the strength of that activity. Five of the grades have a high antimicrobial activity, able to reduce pathogen numbers by 99.99%, while four of the grades have a lower antimicrobial activity, though still able to reduce pathogen numbers by at least 99%. This allows medical device and instrument manufacturers to pick the grade with just the right strength for their specific application: for example, medical devices that are not regularly touched may require grades with lower antimicrobial activity.
SABIC says its range of antimicrobial plastics can be used to produce surgical instruments, monitoring and imaging devices, equipment for delivering fluids and drugs, and durable medical equipment such as beds and operating tables.
The Silver Bullet
Even though these antimicrobial plastics are fairly recent developments, the medical benefits of silver have been known about for over two thousand years. The discovery of chemical antibiotics in the first half of the 20th century caused silver to be sidelined. But the rise of antibiotic-resistance in many bacteria has encouraged a renaissance, as has our ability to produce nanosilver, which both enhances bulk silver’s antimicrobial activity and is easy to incorporate into medical products such as plasters, bandages, and now plastics.
Until recently, however, scientists were unclear about exactly how silver produces its antimicrobial effect. They knew that silver ions derived from bulk silver and nanosilver actually do the damage, but exactly how they damaged the microbes remained unclear. Only in June this year did a paper in Science Translational Medicine provide an answer.
A team of scientists from Harvard University and Boston University in the USA used a variety of analytical and microscopy techniques to investigate what happens when both normal and antibiotic-resistant strains of E. coli are exposed to silver ions. They found that the ions disrupt multiple bacterial processes, including cellular metabolism and iron homeostasis, as well as stimulating the production of reactive oxygen species (ROS) in the cells.
The ROS cause immediate and serious damage to various essential components of the bacterial cell, including its DNA and outer membrane, all of which can lead to cell death. The disrupted bacterial processes, on the other hand, don’t kill the cell, but further weaken its outer membrane, making it leakier and allowing antibiotics to enter the cell. So, as well as physically damaging bacterial cells, silver ions can also make them more susceptible to antibiotics.
Exploring this enhanced susceptibility in more detail, the scientists discovered that a small amount of silver made E. coli between 10 and 1,000 times more sensitive to three commonly used antibiotics: gentamycin, ofloxacin, and ampicillin. Silver also enhanced the effect of antibiotics against biofilms, and it made tough Gram-negative bacteria, which have an additional outer cell wall, susceptible to antibiotics that normally only work against Gram-positive bacteria, which lack this wall.
All this suggests that as well as helping to prevent HAIs, plastics containing silver nanoparticles may also assist with the treatment of existing HAIs by enhancing the efficacy of antibiotics. Presumably, however, this enhancement effect will only work if the antimicrobial plastic is used to produce an instrument or device that is actually inserted into the body.
Another advantage of silver over chemical antibiotics is that it’s harder for bacteria to develop resistance against the physical effects produced by silver ions, but that doesn’t mean it’s impossible. In the journal Small, Australian researchers recently reported their discovery that whereas silver ions were effective at killing E. coli cells, a species of Bacillus seemed to be immune to their effect, able to adapt to the oxidative stress induced by the ions. Indeed, the reduction in E. coli cells caused by silver ions seemed to spur the fast growth of Bacillus cells.
This indicates that the increased use of nanosilver in everything from hand wash to bandages to plastics, especially in hospitals, may well encourage certain bacteria to develop resistance to some of its effects, in the same way they have developed resistance to chemical antibiotics. Physicians and hospital managers need to be aware of this potential danger and watch out for the development of resistant strains.
What probably makes this resistance less problematic than antibiotic resistance is that silver is not being used to treat HAIs, but rather simply to limit their spread. Conventional antibiotics will still be used to treat HAIs and, as a recent Science Translational Medicine paper shows, the combination of antibiotics and silver can be much more potent than antibiotics on their own.
Furthermore, unlike chemical antibiotics, silver has various effects on bacterial cells. So even if bacteria adapt to withstand the damage caused by ROS, they will probably still be susceptible to the disrupted cellular processes that produce leaks in their outer membrane, making them more susceptible to antibiotics. Thus silver will likely remain an effective weapon against HAIs for the foreseeable future.
What’s more is that antimicrobial silver is not the only way that plastics can help to limit the spread of HAIs. Medical instruments and devices are now repeatedly sterilized to prevent them being colonized by bacteria. There are numerous options for sterilizing these instruments and devices, including hot steam, disinfectants such as ethylene oxide, and high-energy radiation such as gamma, beta, and e-beam radiation.
Radiation Sterilization Resistance
The problem is that these sterilization procedures are quite harsh and can damage and discolor instruments and devices made from plastic. So plastic producers have been developing polymers that can withstand repeated application of these kinds of harsh procedures without damage or discoloration.
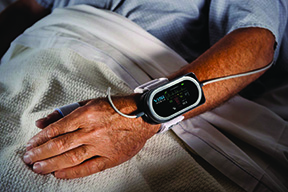
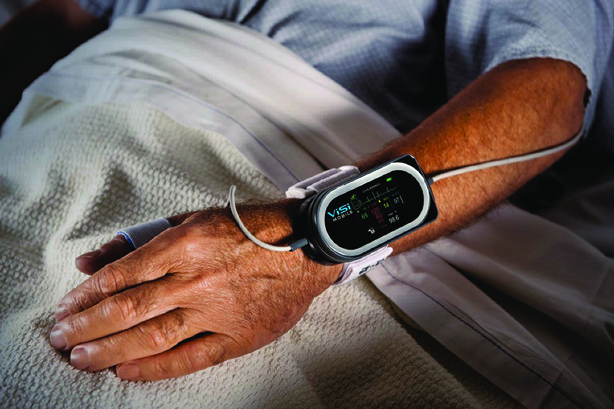
For example, Eastman Chemical has developed its tough, chemically resistant, polyester-based resin, Tritan. According to Eastman, Tritan’s chemical resistance means that medical instruments made from this plastic can be frequently cleaned with strong disinfectants and chemicals without cracking or being discolored. Tritan is already being used by the U.S. medical device company Sotera Wireless in its ViSi Mobile System, a wrist-worn device that monitors vital signs such as heart and pulse rate, blood pressure, oxygen saturation, and skin temperature. Such devices are designed by to be worn repeatedly by different patients and so need to be frequently sterilized.
The other advantage of Tritan is that it’s naturally clear. This means it can be used to produce transparent containers where the objects inside can be seen without opening the containers, thereby reducing the chance of bacterial contamination. For instance, the U.S. company Pevco has used Tritan to produce clear pneumatic tube carriers for transporting small materials such as drugs and laboratory specimens via the tube systems used in many hospitals.
Similarly, the Brazilian medical device company Sartori Instruments has used SABIC’s clear Ultem polyetherimide resin to produce a plastic sterilization box with a transparent cover, allowing physicians to check the contents without having to open the cover.
SABIC has also designed its new Lexan HPS polycarbonate resins to be able to withstand sterilization by gamma radiation, which is an increasingly popular sterilization method for medical instruments as it is fast, inexpensive, and penetrates deeply into the material.
Some plastics producers are also ensuring that their new antimicrobial plastics are robust enough to withstand these sterilization methods. For example, Evonik’s Cyrolite and Cyrex Protect reportedly can withstand sterilization by ethylene oxide and by e-beam and gamma radiation, while BASF says that its HyGentic range can be disinfected using conventional procedures.
A Reason for Some Optimism
The introduction of new hygiene measures do already seem to be stemming the tide of HAIs. Data published by the UK Health Protection Agency in 2012 revealed that the total prevalence of HAIs in English hospitals decreased from 8.2% in 2006 to 6.4% in 2011, with MRSA and C. difficile infections falling by 70% over that period. Similarly, the CDC reported in 2012 that the incidence of life-threatening MRSA infections in U.S. hospitals fell by 48% between 2005 and 2010.
Despite these declines, HAIs still represent a major health risk, as the recent ECDC report makes clear. Hopefully, though, the introduction of medical devices and instruments made from plastics with antibacterial properties that can withstand harsh sterilization methods will allow this downward trend to continue. Then admission to a hospital will be no more stressful and traumatic than it needs to be.