SAMHSA-Compliant LC/MS/MS Analysis of Benzoylecgonine in Urine with Agilent Bond Elut Plexa PCX and Agilent Poroshell 120
Irina Dioumaeva, John M. Hughes
Agilent Technologies, Inc.
Application Note
Forensic Toxicology
Abstract
New guidelines from the US Substance Abuse and Mental Health Services Administration (SAMHSA), effective October 2010, allowed LC/MS/MS methods to be used for confirmation of initial drug tests [1]. LC/MS/MS methods are often less complicated than previously employed GC/MS methods because they do not typically require a derivatization step. We present a method for analysis of benzoylecgonine that meets the most recent SAMHSA guidelines to demonstrate linearity, limit of detection (LOD), accuracy, and precision, as well as measurement of matrix effects, extraction recovery, and overall process efficiency. This is one of a suite of six simplified methods covering all classes of SAMHSA-regulated drugs and using premier Agilent products, including Agilent Bond Elut Plexa PCX mixed-mode polymeric SPE sorbent, Agilent Poroshell 120 EC-C18 2.7 µm superficially porous LC column, Agilent 1200 Infinity LC system, and Agilent 6460 Triple Quadrupole LC/MS system with Agilent Jet Stream Technology (AJST) enhanced electrospray source.
Introduction
Benzoylecgonine (BE) is a major urinary metabolite of cocaine. Cocaine hydrolysis to benzoylecgonine occurs enzymatically (in the liver), as well as without catalysts at alkaline pH [2]. The SAMHSA-established confirmation cutoff concentration for benzoylecgonine is 100 ng/mL, and a LOD at 10% of the cutoff would be 10 ng/mL [1].
The extraction method described in this application note provides reproducible high recoveries of benzoylecgonine due to unique properties of the Agilent Bond Elut Plexa polymer. Unlike other polymeric sorbents, Plexa possesses an amide-free hydroxylated particle surface that excludes protein binding. This results in minimized ion suppression and maximum sensitivity. Fast flow and reproducible performance are due to the narrow particle size distribution with no fines to cause blockages.
A Poroshell 120 EC-C18, 3 × 50 mm,
2.7 µm column was chosen due to its high capacity and excellent separation properties. With superficially porous 2.7 µm particles, Poroshell 120 provides similar efficiency to sub-2 µm UHPLC columns but with about 40% less back pressure, thereby allowing users of even 400 bar LC systems to increase resolution and to shorten both analysis and re-equilibration times by applying a higher flow rate.
With a low sample injection volume of 2 µL and no sample preconcentration, the presented method demonstrates excellent signal-to-noise (S/N) ratios (> 400:1 at
10 ng/mL, 10% of the SAMHSA confirmation cutoff) due to the enhanced sensitivity of the Agilent 6460 Triple Quadrupole LC/MS system with the AJST electrospray source.
Previous methods from Agilent used the Agilent 6410 Triple Quadrupole LC/MS system and other SPE/LC products and procedures [3,4].
Experimental
Analytes
Drug standards were purchased from Cerilliant Corporation as 1 mg/mL (benzoylecgonine) and 100 µg/mL
(benzoylecgonine-D8) solutions in methanol.
Materials and instrumentation
SPE
- Agilent Bond Elut Plexa PCX cartridges, 30 mg, 3 mL
(p/n 12108303) - Agilent vacuum manifold VacElut 20 (p/n 12234100)
- Agilent stopcock valves
(p/n 12234520) - Agilent 2-mL autosampler vials
(p/n 5182-0716) - Agilent screw caps for autosampler vials (p/n 5182-0717)
LC
- Agilent Poroshell 120 EC-C18,
3 × 50 mm, 2.7 µm column
(p/n 699975-302) - Agilent 1260 Infinity LC (G1379B microdegasser, 1312B binary pump in low delay volume configuration, G1367E autosampler, and G1330B thermostat)
MS
- Agilent 6460A Triple Quadrupole LC/MS system with AJS electrospray ionization source
Sample preparation
Pretreatment
Spike 1 mL of urine with ISTD at 200 ng/mL; use of 12 × 75 mm glass tubes is recommended. Add 1 mL of 2% formic acid, vortex; centrifuge if cloudy.
Extraction
- Condition Bond Elut Plexa PCX column with 0.5 mL methanol – soak, then let drip.
- Load sample/supernatants.
- Wash 1: 1 mL 2% formic acid.
- Wash 2: 1 mL of methanol.
- Dry 5–10 minutes under vacuum
(10–15 in Hg). - Elute with 1 mL methanol: ammonium hydroxide (100:20), freshly prepared. Let eluate drip into collection vials, then apply low vacuum (2–3 in Hg).
- Evaporate under a stream of nitrogen to dryness.
- Reconstitute in 1 mL initial mobile phase (10% methanol, 90% water, 0.1% formic acid).
LC/MS/MS
LC conditions
Mobile phase A
0.1% formic acid in water
Mobile phase B
0.1% formic acid in methanol
Flow rate
0.8 mL/min
Gradient
Time (min) % B
0.0 10
0.5 10
2.5 70
2.51 90
5.5 90
5.51 10
Stop time
5.6 min
Post time
2 min
Max pump pressure
400 bar
Injection volume
2 µL
Injection with needle wash
Needle wash
Flush port 75:25 methanol:water for 10 s
Disable overlapped injection
No automatic delay volume reduction
MS conditions
ES source parameters
Ionization mode
Positive
Capillary voltage
3,000 V
Drying gas flow
10 L/min
Drying gas temperature
350 °C
Nebulizer gas
35 psi
Sheath gas flow
12 L/min
Sheath gas temperature
400 °C
Nozzle voltage
0 V
MS parameters
Scan type
MRM
Pre-run script
SCP_MSDiverterValveToWaste()
{MH_Acq_Scripts.exe}
Time segments
#1: 1.2 min - diverter valve to MS
Delta EMV(+)
200 V
Results and Discussion
At acidic pH, the tertiary amine of benzoylecgonine was protonated, and the analyte was efficiently retained on Plexa PCX polymeric sorbent by a combination of hydrophobic interaction and a strong cation exchange.
A 100% methanol wash eliminated most matrix interferences without BE loss from the SPE column. A strong base was added to the organic eluent to break the ionic interaction between the analyte and the strong cation exchange sorbent. Benzoylecgonine recovery was optimized with 20% NH4OH added to methanol shortly before sample elution.
The Poroshell 120 EC-C18, 3 × 50 mm,
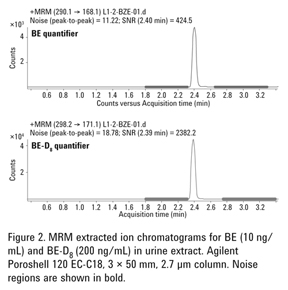
2.7 µm column provided fast separation of benzoylecgonine in urine extract and good peak shape (Figure 2). The LC separation started with a low fraction of the organic solvent (10%) to allow salts and other polar components of urine to elute at the beginning of the sample run. Each sample run started with diverting a first portion of flow (0 to 1.2 minutes) to waste to minimize source contamination. Data collection started at 1.2 minutes, immediately after the diverter valve switch. A flow rate of 0.8 mL/min allowed for short analysis and re-equilibration times.
A S/N ratio >400:1 for the 10 ng/mL peak (Figure 2), upper panel) illustrates a state-of-the-art performance of the Agilent 6460 Triple Quadrupole capable of reliably detecting benzoylecgonine at a small fraction (10%) of the SAMHSA cutoff concentration.
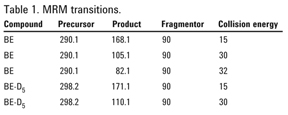
SAMHSA guidelines require one quantifier and at least one qualifier ion for both target compound and ISTD. A third transition for target analyte (Table 1) was provided for additional confidence. The Agilent MassHunter Quantitative software automatically calculated qualifier ion ratios, highlighting those out of acceptable range.
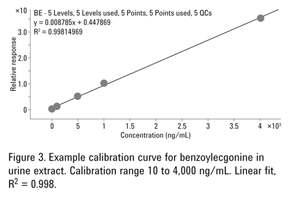
Figure 3 is an example calibration curve for extracted urine standards at five concentration levels of benzoylecgonine. Calibration standards were prepared by spiking negative urine at 10, 100, 500, 1,000, and 4,000 ng/mL. Deuterated internal standard BE-D8 was added at 200 ng/mL. The excellent linear fit with R2 = 0.998 demonstrated linearity of the method across a broad dynamic range of concentrations, as required by SAMHSA guidelines.
Normal, rather than dynamic, MRM scan type can be used with this method, because dynamic MRM has no advantages for detection of a single compound.
Method evaluation
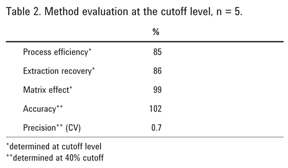
Method performance metrics in Table 2 were calculated according to the principles laid out in Matuszewski et al and widely accepted as an industry standard approach for LC/MS/MS methods [5]. The extraction procedure and the LC/MS/MS measurement were performed for five replicates of negative urine spiked pre-extraction at the cutoff level, and five replicates of negative urine extract reconstituted in initial mobile phase and then fortified at 100 ng/mL with benzoylecgonine (spiked post-SPE ). The third measurement was of initial mobile phase (the reconstitution solvent) fortified to correspond to the cutoff concentration of 100 ng/mL in urine (spiked mobile phase).
Process efficiency (absolute recovery) is a ratio of a peak area of target analyte in urine sample spiked pre-SPE to its peak area in matrix-free spiked mobile phase. Extraction recovery is a ratio of a peak area of target analyte in urine extract spiked pre-SPE to its peak area in an extracted negative urine sample spiked post-SPE. Matrix effect is a ratio of a peak area of target analyte in urine spiked post-SPE to its peak area in spiked mobile phase.
Accuracy is a ratio of a measured concentration calculated using the calibration curve to the expected concentration in a sample spiked with a known amount of target analyte. Precision or coefficient of variation (CV) is a measure of reproducibility and is calculated as a percent standard deviation over the mean of the five measurements.
Table 2 shows high extraction recovery for benzoylecgonine (86%) together with excellent accuracy (102%) and precision (0.7%). Matrix effect of 99% indicates minor ion suppression of a signal due to matrix interferences (1%), thus, confirming an exceptional cleanliness of Plexa PCX-processed extracts.
Conclusions
The solid phase extraction procedure coupled with LC/MS/MS detection method described in this application note is SAMHSA-compliant and provides accurate, precise, and reproducible results for forensic toxicology or other analytical environments with similar requirements for legally defensible data. The hardware setup is the same as in the other 2011 SAMHSA methods from Agilent. These methods are intended for all users of Agilent 1100 and Agilent 1200 Series LCs because the back pressure in the LC system does not exceed 400 bar. Source parameters can be easily modified to use this method with other models of Agilent Triple Quadrupole LC/MS/MS instruments. Electronic copies of the LC/MS/MS acquisition and quantitation methods are available from Agilent Technologies.
References
- SAMHSA (2010) “Manual for Urine Laboratories”, National Laboratory Certification Program, 1 October 2010. U. S. Department of Health and Human Services.
- R. Baselt (2008) Disposition of Toxic Drugs and Chemicals in Man. 8th edition. Atlas Books, Ashland, OH, USA.
- P. Moorman and J. Hughes (2010) “Benzoylecgonine in Urine by LC/Triple Quadrupole Mass Spectrometry (LC/MS/MS)”. SOP, Agilent Technologies, Inc. Publication number 5990-5866EN.
- J. Hughes and P. Moorman (2011) “Confirmation by Triple Quadrupole LC/MS/MS for HHs-compliant Workplace Urine Drug Testing”. Agilent Technologies, Inc. Seminar available from www.agilent.com/chem.
- B. K. Matuszewski, M. L. Constanzer, and C.M. Chavez-Eng (2003) “Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC‑MS/MS”. Analytical Chemistry, 75: 3019-3030.
For More Information
These data represent typical results. For more information on our products and services, visit our Web site at www.agilent.com/chem.
©Agilent Technologies, Inc., 2013
February 15, 2013
View this Application Note in its entirety: 5990-9624EN